In the engine tuning world, detonation is defined as one of the following: combustion that causes engine damage; combustion that causes banging or pinging noises; or combustion that causes power loss, bucking, or kicking. Detonation is not controlled and often unwanted. It occurs when fuel in the cylinder auto-ignites outside the intended flame front of the spark ignition.
Detonation does not always cause damage. At lower engine loadings during part throttle or low RPM, detonation may be warranted. For example, during the late ’70s and ’80s, pinging during normal operation was common with carbureted engines. Certain intake manifold design compromises combined with smog equipment caused lean fuel mixtures that burned outside of the controlled flame front from the spark plug.
Minor detonation sometimes occurs that is not heard through mufflers at low load or even with loud open exhaust. Major detonation causes a more severe noise during engine loading where the throttle is open and the engine is twisting hard against a heavy load.
Detonation and Preignition
Preignition is auto-ignition of the air/fuel mixture before the spark plug fires. The auto-ignition occurs at a location in the cylinder outside of the controlled flame front from spark ignition.
Similarly, detonation is auto-ignition of fuel, usually after the spark plug fires. Like preignition, detonation occurs outside of a controlled flame front from the spark plug. The term detonation is often used by racers as both preignition (before the spark), as well as uncontrolled burning after the spark. The same convention is used in this article.
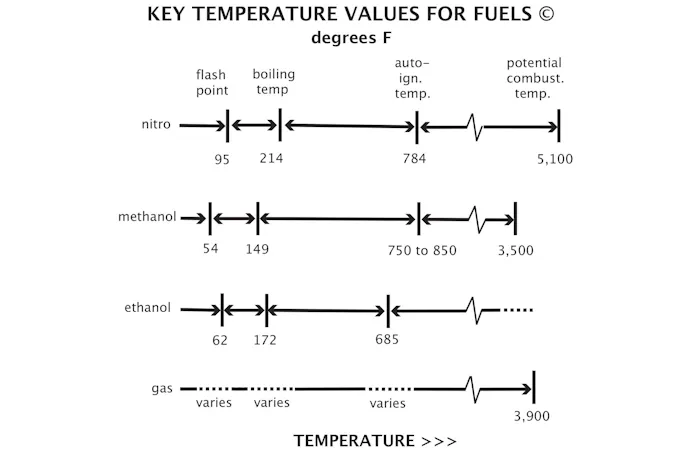
Both preignition and detonation are from auto-ignition of fuel. They share characteristics — like a very high burn speed — which are comparable to explosive flame speeds. These include firearms muzzle velocities or explosives combustion velocities — typically well over 1,000 feet per second. The high velocity causes noise due to pressure fronts that collide within the cylinder.
Detonation and RPM
Detonation can be masked at higher RPM by high frequency noise, like that of an exhaust valve opening. It can be such a brief occurrence that it doesn’t cause damage before the exhaust valve opens, relieving the cylinder pressure and ending the detonation.
At lower engine speeds the time between detonation and opening of the exhaust valve is a longer interval, so detonation is more noticeable. As the RPM increases, it may sound like the detonation goes away because of shorter intervals between detonation and opening of the exhaust valve.
Racing engines in the ’30s and ’40s ran on lower octane gasoline, as higher octane gasoline was not yet developed. The lower octane fuels were susceptible to detonation as racers raised engine compression ratio for more power. Detonation was especially detectable at low engine speeds. To combat the low-speed detonation, those early racing engines were revved continuously at higher engine speeds to inhibit the effects of detonation.
If the engine was lugged by mistake, detonation could cause poor performance and possible engine damage. As a result, drivers coming into the pits for service would free-rev their engines continuously. Slipping the clutch to launch from the pits became an art form for many successful racing drivers. When starting in the pits, there was a great risk of engine stalling from a combination of inadequate clutch slipping, low engine torque at low-RPM, and detonation at low-RPM.
The best performance for modern day gasoline-fueled engines is achieved with a racing gasoline blend with an octane rating just high enough to avoid detonation. A gasoline blend with a higher octane rating does not usually increase performance by itself. Instead, the slower burn rate of high octane gasoline will often actually reduce an engine’s performance without other changes made to take advantage of the higher octane.
Gasoline’s octane need is characteristic of a specific RPM operating range. If that range is changed, a racing gasoline with a different octane may be needed. For example, if the engine spends more time under load at a lower engine speed, the engine may encounter detonation, whereas it would not detonate at the same load higher in the RPM range. A racing gasoline with a higher octane may be needed to combat the potential for detonation operating in the lower RPM range.
Dissociation From Combustion
Fuels dissociate, or break down, into different intermediate chemicals during compression, heating, and combustion. These intermediate chemicals can alter the auto-ignition temperature of the mixture from that of the original fuel alone. Many times, the wrong tune-up call is made because of detonation, assuming data based on just the properties of the primary fuel, when auto-ignition temperature changes from dissociation should have been taken into account.
In blown alcohol drag racing, competitors with higher static compression usually have to run a richer mixture than those with lower static compression to inhibit detonation. However, there is a point at which the need for additional enrichment tapers off. One competitor reported that beyond a certain point in compression increases, further enrichment was not needed, while the engine made more power with more compression. He continued to raise compression further and achieved national record-running performance. At a certain point, the extra-high compression was actually preventing the formation of detonation-sensitive dissociates.
Dissociate Causes of Detonation
With different racing fuels, some of the previously described dissociate formations can be more prone to detonation than others. The tune-up can affect the compression and heating which will affect what dissociates are formed, even with the same fuel. These dissociates then affect the detonation sensitivity. Additionally, air density changes affect the tune-up which, once again, affects dissociation in a vicious cycle.
For example, changing the closing point of the intake valve in a spark ignition racing engine will change the effective dynamic compression. Changing the compression changes the adiabatic heating and pressure from compression. Sensitivity towards or away from detonation can be triggered by something as simple as a camshaft change or even just retarding or advancing camshaft timing.

Pressure Changes Causing Ignition
Pressure changes the auto-ignition temperature of both the fuel and fuel dissociates, which can initiate detonation. An auto-ignition temperature of a fuel dissociate may be lower than the auto-ignition temperature of the fuel before it breaks down, which can be confusing, when looking at the data for the fuel alone.
During compression, say a mixture of air and dissociated fuel is below the auto-ignition temperature. The pressure wave generated in the cylinder can inhibit this mixture from igniting. However, as the pressure wave goes through the cylinder, it can trigger a change in the auto-ignition temperature of the mixture. Auto-ignition can follow as the pressure wave passes due to the accompanying drop in auto-ignition temperature, strictly from the chemical sensitivity change. Additionally, changes in the cylinder head from piston squish or unshrouding an intake valve can change the pressure wave formation and affect the combination’s overall detonation sensitivity.

Dissociation With Different Fuels
Gasoline
According to writings by the late Harry Ricardo (The High-Speed Internal-Combustion Engine, 3rd Edition, Blackie & Son Limited, 1950), who was a combustion engineering expert, unstable peroxides are formed as intermediate dissociates during gasoline combustion, which happen to be very prone to detonation. Tetra-ethyl lead is a reactive metallic addition that suppresses detonation from these unstable peroxides. In addition, various gasoline fuel constituents used in common blends have different dissociation properties to help combat the formations of the unstable peroxides. Examples of these constituents used are pentane, hexane, and toluene.
Fuel blending in modern gasolines is done to achieve detonation resistance, among other characteristics. In many racing gasoline brands, several are also blended with tetra-ethyl lead for the same purpose. Other characteristics such as chemical stability, ease of vaporization so that the engine can be started, and manufacturing cost, often limit blend additives and ratios. Those constraints can compromise the detonation resistance capability of some gasoline brands over others under given circumstances. The ideal result is a best-fit mixture or blend to fulfill the particular racing requirement, and why so many different racing gasoline variations exist.
Gasoline blends sold at the pump most often have seasonal changes in blend ratios and fuel content. Winter gasoline is blended for ease of starting, while summer gasoline blends are designed for vapor-lock avoidance. Different seasonal blends will change the dissociation and detonation characteristics, and needs to be accounted for in a performance application. Pump gasoline purchased in one season may encounter detonation issues if it is run in another season, due to the variance in blend.
Ethanol Gasoline Blends (E85)
E85 is predominantly (85-percent) ethanol with a small amount (15-percent) of gasoline added. The ethanol content’s high effective octane rating suppresses detonation in high-compression-ratio racing engine if the air/fuel ratio is rich. That would be a lambda of less than one in the EFI computer world. A rich alcohol fuel mixture also cools the cylinder away from auto-ignition temperatures. These rich air/fuel ratios can be run with a predominant alcohol fuel since alcohol does not foul out a spark plug like other fuels can. However, excessive richness reduces power output, so tuning the air/fuel ratio is vital. On the other end of the specturm, excessively rich mixtures can cool the intake too much, suppressing vaporization and inducing detonation from a vapor lean condition. This is the result of excess fuel condensation from cooling.
Methanol
Methanol as well as ethanol will dissociate into hydrogen and carbon monoxide during compression heating. Methanol and ethanol will also partially dissociate into hydrogen and carbon monoxide during boost in an engine with large enough pressures from forced induction, prior to, and in addition to piston compression. However, compression pressure retards the amount of dissociation that occurs. Therefore heat causes dissociation to go in one direction, and pressure from compression (or boost) causes dissociation to go in another. Combustion is then a combination of hydrogen, carbon monoxide, and any remaining methanol vapor that has not dissociated.

Differences in compression, engine temperature, camshaft timing, and boost in forced induction engines all affect the amount of dissociation that occurs. The amount of dissociation then affects the combustion characteristics of the charge. For example: hydrogen has very low ignition temperature and is more prone to backfiring into the intake, as it doesn’t necessarily need a traditional ignition source. That is often mistaken as detonation when, in fact, it is excess hydrogen dissociation that is reacting.
A tune-up or air density change can change the hydrogen dissociation and make or avoid engine backfiring. When backfiring from hydrogen dissociation occurs, subsequent engine disassembly often does not reveal any engine damage. The variation in auto-ignition temperatures of methanol is due to different amounts of dissociation from the tune-up and air density changes.
Methanol has oxygen in the fuel while traditional gasoline does not. As such, methanol can detonate with less air in the mix than gasoline. An air/fuel weight-ratio of 8-to-1 would be overly rich for gasoline and not detonate but could detonate with methanol fuel. That threshold changes with oxygen variations in the air from air density changes.

Nitromethane
Nitromethane dissociates in different phases. For a brief moment, some of these phases are sequential, and some are even simultaneous during the ignition and combustion process. However, many phases of nitromethane dissociation occur simply from compression heating and combustion.
The first phase is endothermic. It absorbs heat and acts like it is hard to ignite. This is why a magneto ignition with a long spark dwell time is more effective with nitromethane fuel mixtures to get past the first dissociation phase of combustion. The second and remaining dissociation phases of nitromethane combustion can be exothermic, that is, burning and giving off heat (Chem-Supply Material Safety Data Sheet, nitromethane, 1CHOP, December, 2000).
Multiple dissociation phases occur during combustion with different intermediate compounds and with different auto-ignition (detonation) characteristics. Different mixtures of nitromethane and methanol add further complexity to detonation sensitivity changes since methanol has its own set of dissociates and behaviors. As a result, the tuning directions can be troublesome and inconsistent from run to run.
Some nitro tune-ups may be more prone to detonation when leaning the mixture (higher air/fuel ratio). Some nitro tune-ups may be more prone to detonation with enrichment of the mixture (lower air/fuel ratio). The best tune-up routine is to make as few changes as possible to engine compression, boost, fuel mixture, fuel temperature, and others to dial-in engine power for the performance envelope. Making multiple changes from run to run makes it almost impossible to get control over the tune-up, because of that wandering auto-ignition temperature characteristic. Major engine failures can occur as a result.

Air/Fuel Ratio Changes
Changes in air/fuel ratio will also change the auto-ignition sensitivity characteristics. This change is complex depending on the amount of enrichment. Enrichment up to a point tends to decrease the auto-ignition sensitivity. Enrichment in methanol or ethanol can cool the cylinder temperature to where the engine will not detonate. Excess enrichment beyond a certain air/fuel ratio with those fuels can increase the auto-ignition sensitivity however. By causing excessive cooling and fuel condensation out of the inlet air charge, a vapor-lean condition is created and auto-ignition can occur. It can also slow down the flame speed, extending the combustion event into the exhaust stroke. That can lead to a backfire in the intake when the intake valve opens.
In the other direction, less enrichment beyond a certain optimum air/fuel ratio tends to increase the auto-ignition sensitivity. With methanol or ethanol, less enrichment will not cool the cylinder temperature enough, raising temperature enough to where the engine may then detonate, especially if high compression ratios are used.

Excessive reduction of enrichment may reduce power since there is less fuel to burn. Continued reduction of enrichment beyond a certain point may not detonate since the extreme lean condition results in a shortage of fuel to burn, and flame speed slows down. Somewhere in that lean direction, flame speed can be slowed, continuing past the exhaust stroke. That can then, like excessively rich conditions, cause a backfire in the intake.
Combination Effects are Complex
A nitro-methanol fuel mixture up to about 87-percent nitro, with increased richness, is less prone to detonation. That is the same behavior as most other fuels, especially alcohol fuels. However, a nitro-methanol mixture above 87-percent nitro with increased richness becomes more prone to detonation. That is because of an abundance of excess oxygen in the fuel. That excess oxygen in the higher percentage sensitizes the mixture towards lower auto-ignition temperature. A richer mixture of high percentage nitro mixtures has more excess oxygen and more detonation sensitivity.
If there is one thing to take away from all this, it’s that in a racing environment, the cause of detonation can be a complex issue, and isn’t as simple as “If X happens, then perform Y to fix.” When you’re at this level of performance the number of factors that could be affecting your power-robbing and potentially engine-damaging detonation issue requires a thorough understanding of what is happening with your fuel between the time it is first introduced to the atmosphere and the opening of your exhaust valve.
You might also like
Mercedes-AMG’s M139 Is The Most Powerful 4-Cylinder For 7-Years Running
The M139 engine from Mercedes-AMG debuted as the most powerful 4-cylinder production engine. Now, it retains the title with even more power.





